Designing an effective gRNA depends on several factors, including specificity, efficiency, and compatibility with your CRISPR system. General guidelines include:
- Choose a sequence complementary to your target DNA and containing the appropriate protospacer adjacent motif (PAM) for your Cas nuclease.
- Avoid sequences with high similarity to non-target regions to minimize off-target effects. A good gRNA has a high on-target score and low off-target score, which our design tools rank accordingly.
- Avoid Poly-T sequences (≥4 consecutive Ts) as they terminate transcription
- Minimize hairpin formation in the gRNA.
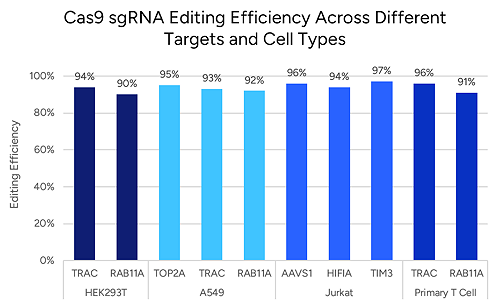
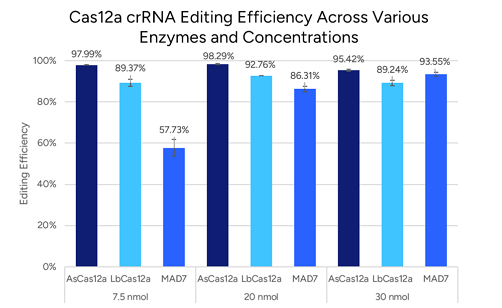
- For Cas9: optimize GC content and spacer length (generally 40–80% GC and 17–24 nucleotides). Cas12a also benefits from moderate GC content, though the optimal range is less strict.
- Base and prime editing require additional considerations:
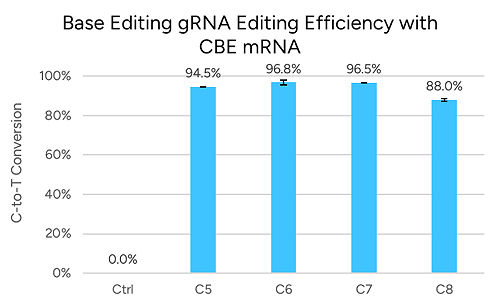
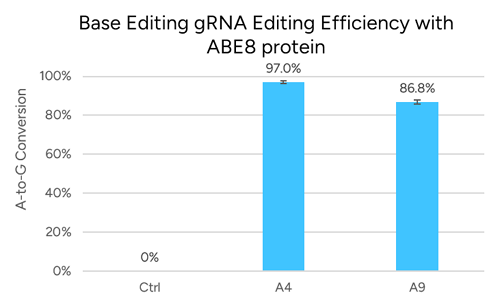
Base editing: the target base must fall within the editing window (~positions 4–8 for cytosine editors, ~4–7 for adenine editors).
Prime editing: pegRNA include a primer binding site (PBS) and a reverse transcriptase template (RTT), each with specific design constraints (PBS length, melting temperature, etc.).
To streamline your design process, you can use our OmniGuide RNA Design Tool to generate Cas9 sgRNA, Cas12a crRNA, or pegRNA, or our HDR Knock-In Design Tool for designing Cas9 or Cas12a guides together with a customized HDR template for knock-in experiments.