-
REAGENT SERVICES
Hot!
-
Most Popular Services
-
Molecular Biology
-
Recombinant Antibody/Protein
-
Reagent Antibody
-
CRISPR Gene Editing
-
DNA Mutant Library
-
IVT RNA and LNP Formulations
-
Oligo Synthesis
-
Peptides
-
Cell Engineering
-
- Gene Synthesis FLASH Gene
- GenBrick™ Up to 200kb
- Gene Fragments Up to 3kb now
- Plasmid DNA Preparation Upgraded
- Cloning and Subcloning
- ORF cDNA Clones
- mRNA Plasmid Solutions New!
- Cell free mRNA Template New!
- AAV Plasmid Solutions New!
- Mutagenesis
- GenCircle™ Double-Stranded DNA New!
- GenSmart™ Online Tools
-
-
PRODUCTS
-
Most Popular Reagents
-
 Instruments
Instruments
-
Antibodies
-
ELISA Kits
-
Protein Electrophoresis and Blotting
-
Protein and Antibody Purification
-
Recombinant Proteins
-
Molecular Biology
-
Stable Cell Lines
-
Cell Isolation and Activation
-
 IVD Raw Materials
IVD Raw Materials
-
 Therapy Applications
Therapy Applications
-
Resources
-
- All Instruments
- Automated Protein and Antibody Purification SystemNew!
- Automated Plasmid MaxiprepHot!
- Automated Plasmid/Protein/Antibody Mini-scale Purification
- eBlot™ Protein Transfer System
- eStain™ Protein Staining System
- eZwest™ Lite Automated Western Blotting Device
- CytoSinct™ 1000 Cell Isolation Instrument
-
- Pharmacokinetics and Immunogenicity ELISA Kits
- Viral Titration QC ELISA Kits
- -- Lentivirus Titer p24 ELISA KitHot!
- -- MuLV Titer p30 ELISA KitNew!
- -- AAV2 and AAVX Titer Capsid ELISA Kits
- Residual Detection ELISA Kits
- -- T7 RNA Polymerase ELISA KitNew!
- -- BSA ELISA Kit, 2G
- -- Cas9 ELISA KitNew!
- -- Protein A ELISA KitNew!
- -- His tagged protein detection & purification
- -- dsRNA ELISA Kit
- -- Endonuclease ELISA Kit
- COVID-19 Detection cPass™ Technology Kits
-
- Automated Maxi-Plasmid PurificationHot!
- Automated Mini-Plasmid PurificationNew!
- PCR Reagents
- S.marcescens Nuclease Benz-Neburase™
- DNA Assembly GenBuilder™
- Cas9 / Cas12a / Cas13a Nucleases
- Base and Prime Editing Nucleases
- GMP Cas9 Nucleases
- CRISPR sgRNA Synthesis
- HDR Knock-in Template
- CRISPR Gene Editing Kits and Antibodies
-
![AmMag™ Quatro Automated Plasmid Purification]() AmMag™ Quatro automated plasmid purification
AmMag™ Quatro automated plasmid purification
-
![Anti-Camelid VHH]() MonoRab™ Anti-VHH Antibodies
MonoRab™ Anti-VHH Antibodies
-
![ELISA Kits]() ELISA Kits
ELISA Kits
-
![Precast Gels]() SurePAGE™ Precast Gels
SurePAGE™ Precast Gels
-
![Quatro ProAb Automated Protein and Antibody Purification System]() AmMag™ Quatro ProAb Automated Protein and Antibody Purification System
AmMag™ Quatro ProAb Automated Protein and Antibody Purification System
-
![Target Proteins]() Target Proteins
Target Proteins
-
![AmMag™ Quatro Automated Plasmid Purification]() AmMag™ Quatro automated plasmid purification
AmMag™ Quatro automated plasmid purification
-
![Stable Cell Lines]() Stable Cell Lines
Stable Cell Lines
-
![Cell Isolation and Activation]() Cell Isolation and Activation
Cell Isolation and Activation
-
 IVD Raw Materials
IVD Raw Materials
-
![Quick
Order]() Quick Order
Quick Order
-
![Quick
Order]() Quick Order
Quick Order
- APPLICATIONS
- RESOURCES
- ABOUT US
- SIGN IN My Account SIGN OUT
- REGISTER
Resources » Weekly Scientific Insight » Unlocking Affordable, Scalable Therapies with Allogeneic Engineering and Advanced Gene Editing
scott.pritchett
Unlocking Affordable, Scalable Therapies with Allogeneic Engineering and Advanced Gene Editing

Author: Dr. Lumeng Ye, Director of Novel Therapeutic Materials
August 19, 2025
Since the FDA approvals of Kymriah and Yescarta in 2017, more than 10 autologous CAR-T therapies have been approved globally to treat B-cell lymphoma and multiple myeloma. More recently, new modalities, such as TCR-T for synovial sarcoma and TIL therapies for advanced melanoma, have also received FDA approval. Despite these remarkable therapeutic achievements, CAR T-cell treatments face significant challenges, including systemic toxicities, limited applications in hematologic cancers, and complex, expensive manufacturing processes.
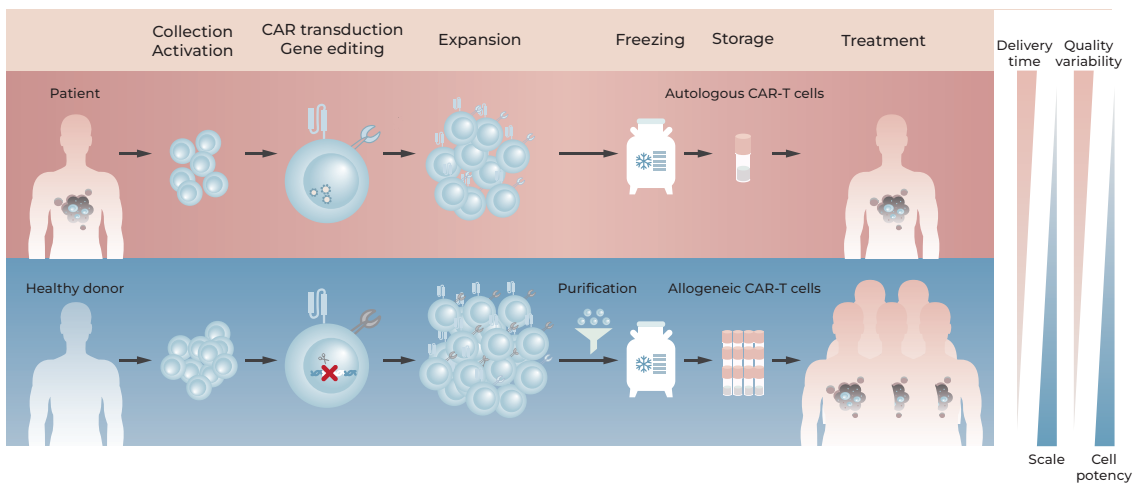
To address these challenges, transitioning CAR-T manufacturing from an autologous approach to an allogeneic approach is essential. Allogeneic manufacturing eliminates patient-specific steps such as lympho-depletion, blood collection, personalized CAR-T production, and QC testing. This streamlined method not only dramatically shortens the treatment process but also ensures timely delivery, reducing the risk of manufacturing failure due to poor quality or insufficient immune cells from individual patients. This transition holds the potential to enhance scalability, improve success rates, and significantly reduce the overall cost of CAR-T therapies. The following figure illustrates the comparison between autologous and allogeneic CAR-T cell therapies.
Advancing Allogeneic Therapies Through Gene Editing
While early attempts at allogeneic immune cell therapies for cancer struggled with issues such as poor persistence after transfusion, advancements in gene editing present a promising solution. For autoimmune diseases, allogeneic universal CAR-T therapies have demonstrated efficacy across multiple indications, such as systemic lupus erythematosus (SLE), idiopathic inflammatory myopathies (IIM), severe myositis, and systemic sclerosis (SSc). Engineering these therapies involves multiplex genome editing, often requiring simultaneous gene knockouts and knock-ins, which can now be achieved more efficiently using non-viral processes, reducing costs and minimizing safety risks.
During therapy design, CMC optimization, and clinical validation processes, new gene editing tools supporting the simultaneous modification of multiple genetic targets are being developed. These advancements enable identification of novel targets to prevent CAR-T exhaustion, enhance persistence, and improve adaptation within the tumor microenvironment through functional genomics screening studies and comprehensive sequencing of edited cells. These tools will support the creation of universal allogeneic CAR-T therapies. With continuous improvement, these therapies could eventually replace autologous CAR-T treatments for hematologic cancers and solid tumors.
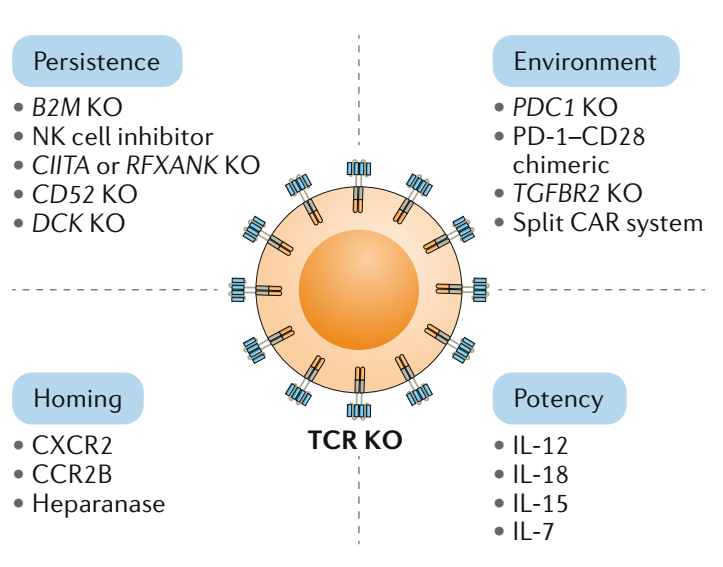
Source: S. Depil, P. Duchateau, S. A. Grupp, G. Mufti & L. Poirot. ‘Off-the-shelf’ allogeneic CAR T cells: development and challenges. Nature Reviews Drug Discovery volume 19, 2020
Optimizing CAR-T Persistence and Precision
A key strategy to prevent CAR-T exhaustion is moderating the density of CAR expression on T cells by controlling the insertion copy number per cell and the expression levels of each CAR gene. Using CRISPR-HDR technology, the CAR sequence can be precisely inserted into the desired reading frame with one or two copies per cell, driven by the endogenous promoter for optimal expression. Recent advancements, such as Cas-targeting sequence (CTS) modifications for CRISPR nucleases like Cas9 and Cas12, have further improved HDR efficiency. Innovative methods like SLEEK (Selection by Essential-gene Exon Knock-in) and SEED (Synthetic Exon/Expression Disruptors) leverage advanced counter-selection strategies to achieve nearly 90% purity in engineered CAR-positive T cells in final therapeutic products, meeting standards for drug purity and homogeneity.
Design Innovations to Improve Potency and Minimize Side Effects
Functionality by design, akin to Quality by Design principles, is rapidly evolving and is critical for delivering next-generation CAR-T therapies. Third-generation CAR-T therapies demonstrate superior proliferation and persistence in vivo compared to second-generation counterparts. The co-stimulatory domains within CAR constructs significantly affect the metabolism and fate of CAR-expressing T cells. For example, the 4-1BB co-stimulatory domain has been shown to provide superior cytokine release while mitigating T-cell exhaustion triggered by scFv-induced CAR aggregation and tonic signaling. Advanced CAR designs incorporating one or more highly sensitive and specific binding domains, combined with the appropriate co-stimulatory domains and even regulatory logic switches, can balance potency and persistence to deliver more effective therapies with reduced side effects. These complex CAR designs can be introduced into T cells via non-viral, targeted genomic editing methods.
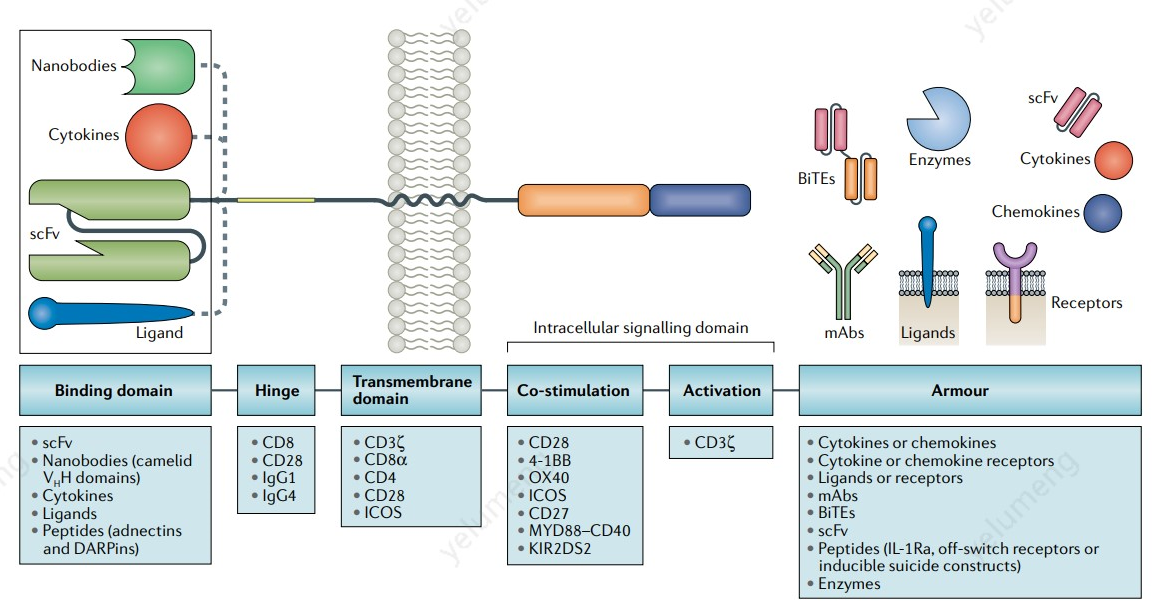
Source: Sarwish Rafiq, Christopher S. Hackett & Renier J. Brentjens. Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nature Reviews Clinical Oncology volume 17, 2020
By combining advanced gene editing tools into an integrated workflow, researchers can efficiently generate powerful antigen-specific treatments with enhanced persistence during therapy. Leveraging therapeutic designs using healthy donor-derived immune cells enables large-scale manufacturing with improved editing efficiency and enhanced cell proliferation rates. Ultimately, this approach can significantly reduce the per-patient cost of CAR-T and other gene therapies while ensuring faster and more effective treatments. Eliminating lengthy manufacturing waiting times also allows patients to access therapies earlier, preventing further progression of malignant cancers.
Learn more about our CRISPR gene editing solutions here.
Note from the editor: If you have any questions about this blog or other interesting topic suggestions related to gene editing or gene and cell therapy for future posts, please reach out at [email protected].
Subscribe to have the latest weekly scientific insights delivery to your inbox!
* We'll never share your email address with a third-party.

-